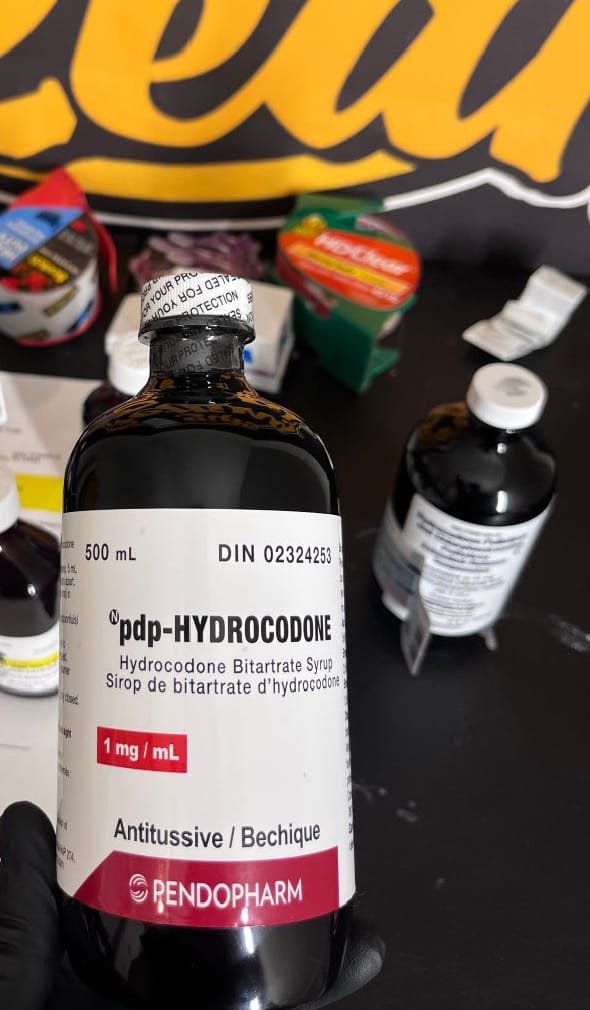
pdp hydrocodone cough syrup
$300.00 – $5,500.00Price range: $300.00 through $5,500.00
pdp hydrocodone cough syrup
PDP hydrocodone cough syrup is a prescription-only liquid medication formulated to suppress severe, persistent cough. The abbreviation “PDP” in pharmaceutical labeling typically refers to the distributor or packaging designation rather than a different active chemical compound. The therapeutic effect of PDP hydrocodone cough syrup comes from hydrocodone, an opioid agonist with clinically recognized antitussive (cough-suppressing) properties. Because it contains an opioid, this medication is regulated as a controlled substance in many jurisdictions and is dispensed strictly under medical supervision.
From a clinical standpoint, PDP hydrocodone cough syrup is generally reserved for short-term management of nonproductive cough that has not responded adequately to non-opioid therapies. Physicians may consider it when a cough significantly disrupts sleep, causes chest discomfort, or interferes with recovery from an acute respiratory illness. It is not intended for routine cough associated with mild colds, nor is it designed for chronic, untreated pulmonary conditions without diagnostic evaluation.
pdp hydrocodone
Hydrocodone is a semi-synthetic opioid derived from codeine. In pdp hydrocodone, it functions primarily as a centrally acting antitussive. The drug binds to opioid receptors in the brainstem, specifically within the medullary cough center, thereby elevating the threshold required to trigger the cough reflex. This mechanism reduces the frequency and intensity of coughing episodes.
In some formulations, hydrocodone may be combined with an antihistamine (such as homatropine or chlorpheniramine) or other adjunctive agents, depending on the product configuration. The combination approach is designed to address both the neural cough reflex and associated upper respiratory symptoms. However, the defining pharmacological feature of PDP hydrocodone cough syrup remains its opioid-mediated suppression of cough.
hydrocodone pdp
Healthcare providers prescribe hydrocodone pdp primarily for short-term relief of severe, dry (nonproductive) cough related to upper respiratory tract infections, bronchitis, or other acute inflammatory conditions. It is not indicated for productive coughs in which mucus clearance is clinically beneficial, as suppressing such a cough may impede airway clearance and prolong illness.
Due to the respiratory depressant properties of opioids, hydrocodone-containing cough syrups are typically not recommended for children and must be used cautiously in elderly patients. The prescriber evaluates factors such as respiratory status, concurrent medications, liver function, and history of substance use before initiating therapy.
pdp hydrocodone syrup
As an opioid-containing medication, pdp hydrocodone syrup is subject to strict regulatory control. Pharmacies must comply with controlled substance dispensing protocols, and prescriptions often cannot be refilled without renewed authorization. The regulatory classification reflects hydrocodone’s potential for misuse, dependence, and diversion.
Patients using PDP hydrocodone cough syrup should avoid alcohol, benzodiazepines, sedative-hypnotics, and other central nervous system depressants unless specifically directed by a healthcare professional. Concurrent use of multiple depressant agents significantly increases the risk of profound sedation, respiratory depression, hypotension, and potentially fatal overdose.
| quantity | 1 pint, 5 pint, 10 pint, 25 pint |
|---|
Be the first to review “pdp hydrocodone cough syrup” Cancel reply
Related products
CODEINE SYRUP
CODEINE SYRUP
CODEINE SYRUP
CODEINE SYRUP
CODEINE SYRUP
CODEINE SYRUP
CODEINE SYRUP
CODEINE SYRUP














Reviews
There are no reviews yet.